A HosCom International 2024 Vol. 1 Article
World Information
Risk Factors for Postprandial Hyperglycemia and the Use of Low-GI Foods
Authors:
Dr. Yuji Murata, SARAYA Co., Ltd.
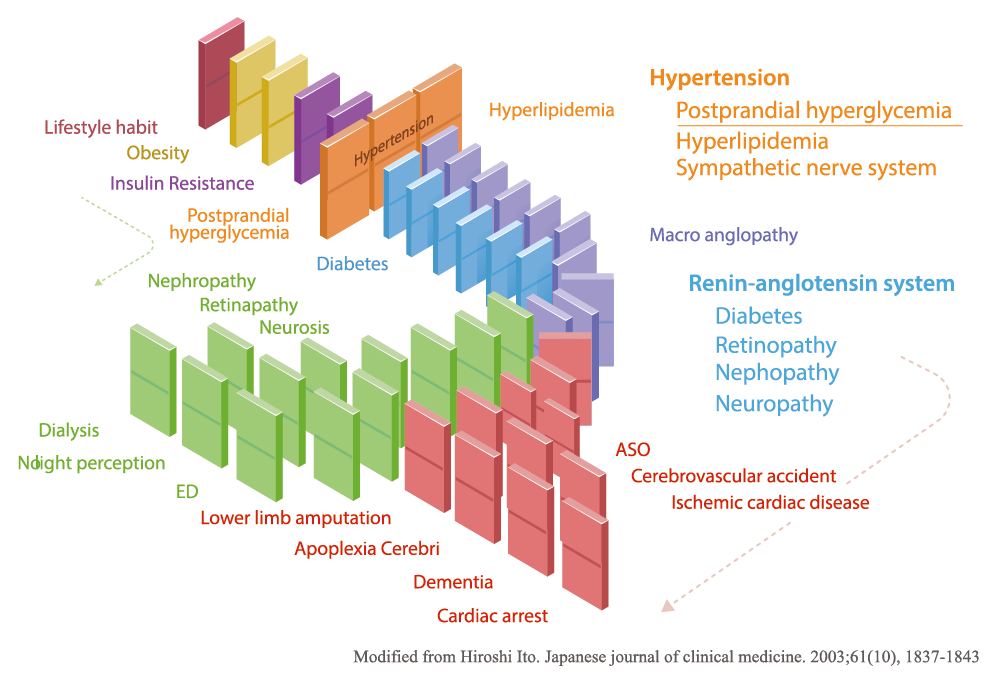
While Japan has the world’s longest life expectancy, its incidence of cardiac and vascular diseases triggered by lifestyle-related diseases is increasing. This effect, “metabolic dominoes” (Figure 1), has been introduced in many publications. If one continues to have inappropriate lifestyle habits, the dominoes of obesity, hyperglycemia, hypertension, lipid disorders, etc., will fall and eventually lead to metabolic syndrome1.
When these metabolic dominoes topple over, they can cause life-threatening diseases, such as angina pectoris, myocardial infarction, and stroke. Therefore, it is important for individuals to mitigate or prevent the development of lifestyle-related diseases in their early stages. Diabetes, dyslipidemia, hypertension, and other lifestyle-related diseases are known as “silent killers” because they progress with few or no subjective symptoms. In other words, the increase in cardiovascular disorders is not due solely to hypertension and lipid abnormalities; it can also be attributed to the lack of awareness of postprandial glycemic control.
In particular, prediabetics and those who require specific health guidance due to higher blood glucose levels than normal often have fasting blood glucose levels below 110 mg/dL—the normal range in Japan. If only fasting blood glucose levels are measured, postprandial hyperglycemia may be overlooked. Even if a patient should undergo a thorough physical examination, the postprandial blood glucose test (oral glucose tolerance test, OGTT) is generally not performed, which can lead to the development and progression of unnoticed diabetes.
Postprandial hyperglycemia causes a decrease in the amount and hampers the function of insulin secretion (insulin resistance), resulting in the inability of body tissues to adequately process glucose, which leads to impaired glucose tolerance. This state of impaired glucose tolerance tends to promote atherosclerosis. Furthermore, as arteriosclerosis progresses, the risk of macrovascular complications such as stroke increases, ultimately resulting in death. For this reason, it is important to manage not only fasting blood glucose levels but also postprandial blood glucose. It has also been reported that abnormal postprandial blood glucose levels worsen when HbA1c exceeds 6.5%. Therefore, improving postprandial hyperglycemia in its early stages and maintaining an appropriate HbA1c levels will reduce the onset and progression of diabetes and elevated fasting blood glucose levels.
In the past, fasting blood glucose levels were used mainly to control health. In recent years, it has been found that a rapid postprandial rise in blood glucose levels contributes to impaired blood flow and vascular damage, which is deeply related to arteriosclerosis and other vascular lesions2,3. The relationship between postprandial hyperglycemia and vascular lesions can be compared to a road with a high volume of traffic that may be severely damaged and in need of repair. The greater the amount of sugar, carbohydrates, and lipids flowing through the blood vessels, the more likely the vascular cells are to become damaged.
Blood vessels are composed of three layers: intima, media, and adventitia. The intima is in contact with the blood circulation in the intravascular lumen and is composed of endothelial cells, a thin layer of cells that make up the inner surface of the vessel. It has been found that vascular endothelial cells constantly produce nitric oxide (NO) and exhibit vascular sclerosis inhibitory effects, vasodilatory effects, and anti-arteriosclerotic effects, thus preventing the vascular wall from developing atherosclerosis. However, when high blood glucose and lipids are present after a meal, these endothelial cells are damaged, the protective action of blood vessels is reduced, and the intima thickens and hardens, leading to arteriosclerosis. The development of arteriosclerosis further leads to the clogging (infarction) of blood vessels, thus resulting in cerebral infarction, myocardial infarction, and other life-threatening conditions.
In response to this, SARAYA provided the world with LAKANTO®, a zero-calorie sugar-substitute sweetener made by adding monk fruit extract to erythritol4. LAKANTO® is a vegetative sweetener that undergoes thorough quality control and does not affect blood sugar levels or insulin.
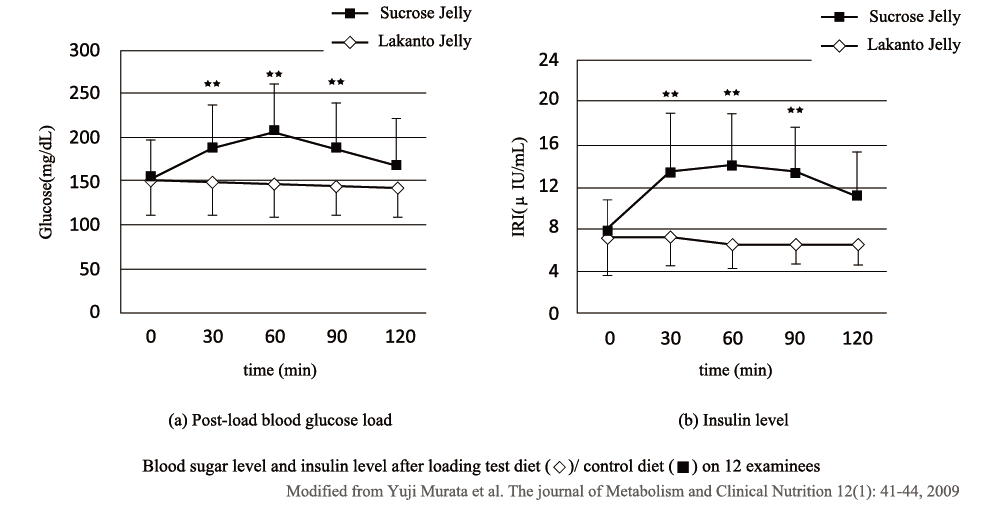
Figure 2 shows the effects of LAKANTO® on blood glucose and insulin in diabetics. Prototype jellies containing 25 g of LAKANTO® (test meal) and jellies containing 25 g of sucrose (control meal) were consumed by 12 diabetic patients, and blood glucose levels were measured at 30-minute intervals up to 120 minutes after consumption. The results of these clinical studies confirmed that LAKANTO® had absolutely no effect on blood glucose and insulin levels.
As derivative products of LAKANTO®, SARAYA launched seasonings including “LAKANTO® Sukiyaki Sauce” and “LAKANTO® Low Carb All-purpose Vinegar” as well as “Healthy Rice,” a low Glycemic index (GI) food that suppresses the rapid rise in blood glucose after meals. “Healthy Rice,” which can be eaten as a staple food, consists mainly of special barley, which is rich in dietary fiber and does not easily raise blood sugar, combined with high-amylose rice. This product is the world’s first rice designed to prevent a rapid rise in blood glucose levels after consumption (patent registered No. 5614982).
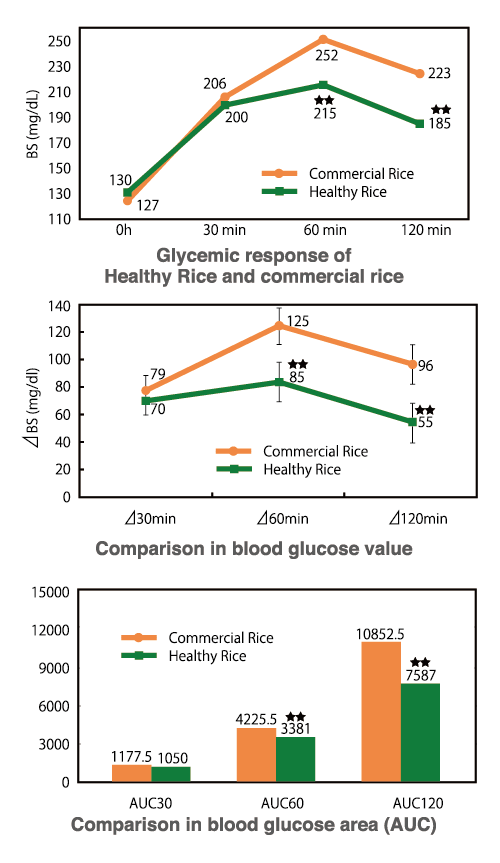
Figure 3. Glycemic responses of “Healthy Rice” compared to commercial rice
Modified from Aya Maeda et al. JOURNAL OF JAPANESE SOCIETY OF CLINICAL NUTRITION. 2012; 34(2).2-7.
|
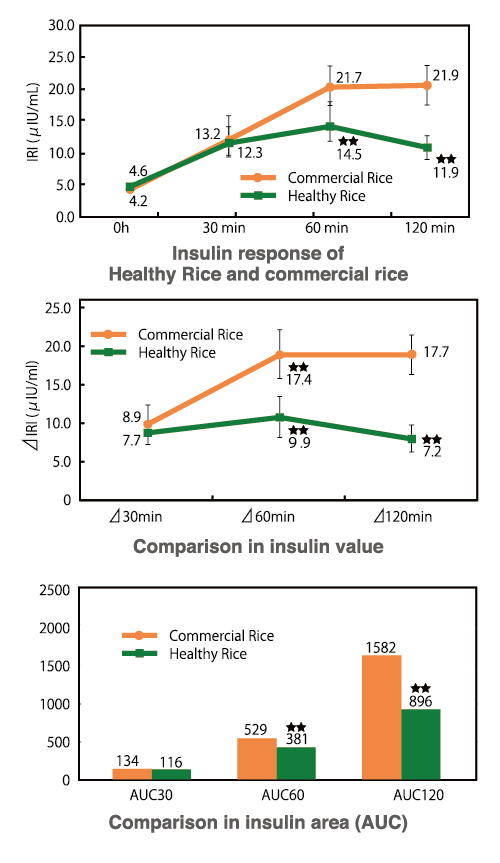
Figure 4. Insulin responses of “Healthy Rice” compared to commercial rice
Modified from Aya Maeda et al. JOURNAL OF JAPANESE SOCIETY OF CLINICAL NUTRITION. 2012; 34(2).2-7.
|
Figure 3 shows the blood glucose levels of 10 Type 2 diabetic subjects (six males and four females) 120 minutes after consuming cooked “Healthy Rice” (test meal) and commercial rice (control meal). The blood glucose levels of individuals who had just consumed “Healthy Rice” (test meal) were statistically significantly lower than those who had just had commercial rice (control meal) in terms of blood sugar (BS), blood sugar elevation (△BS), and blood glucose area (area under the curve, AUC). Figure 4 compares insulin concentrations after consumption of commercial rice (control meal), as in Figure 3. Insulin concentrations after consumption of “Healthy Rice” (test meal) were lower than those of commercial rice (control meal) in terms of insulin value (IRI), insulin elevation value (△IRI), and insulin area (AUC), confirming that there are statistically significant differences between the two types of rice6.
Thus, we find that the food products provided by SARAYA are designed with formulations that do not cause a rapid rise in blood glucose or insulin secretion after ingestion (low-GI foods). We hope that these products will help prevent the progression of lifestyle-related diseases and encourage individuals to take postprandial blood glucose management into consideration andprevent the dominoes of lifestyle-related diseases from toppling over.
-
Publication Date:March 29, 2024
-
Category:Nutrition
HosCom International 2024 Vol. 1

Other Articles in this volume
References
- Ito Hiroshi, メタボリックドミノとは生活習慣病の新しいとらえ方一 <translation> Metabolic Domino - A New Way to View Lifestyle-related Diseases. Japanese Journal of Clinical Medicine. 2003; 61(10): 1837–1843
- David J.A. Jenkins et al. Glycemic index of foods: a physiological basis for carbohydrate exchange. The American Journal of Clinical Nutrition. 1981; 34: 362–366
- Christian K. Roberts et al. Effects of Glycemic Load on Metabolic Health and Type 2 Diabetes Mellitus. Journal of Diabetes Science and Technology. 2009 Jul; 3(4): 697–704.
- Yuji Murata et al. 羅漢果配糖体の甘味特性およびその改善 <translation> Sweetening properties of monk fruit glycosides and their improvement. Food Science and Technology Research. The Japanese Society for Food Science and Technology. 2006; 53(10), 527–533
- Keiko Kondo et al. 糖尿病患者における甘味料「ラカントS」の血糖値、インスリン値に及 ぼす影響 <translation> Effect of the sweetener "Lakanto S" on blood glucose and insulin levels in diabetic patients. The Journal of Metabolism and Clinical Nutrition. 2009; 12(1): 41–49
- Aya Maeda et al. 高アミロース米配合の低GI米による血糖抑制とその意義 <translation> Glycemic control by low GI rice blended with high amylose rice and its significance. Journal of Japanese Society of Clinical Nutrition. 2012; 34(2): 2–7.