A HosCom International 2024 Vol. 3 Article
World Information
Efficacy Testing of Airborne Surface Disinfection Procedures According to EN17272
Authors:
Dr. Florian H. H. Brill, Mr. Henrik Gabriel, Dr. Dajana Paulmann
Dr. Brill + Partner GmbH, lnstitut fur Hygiene und Mikrobiologie
Background
For over 100 years, airborne disinfection processes have been employed as an alternative to complement manual surface disinfection methods involving spraying or wiping1. The advantage of these airborne methods is that they can potentially reach all of the surfaces in a room. Recently, developments have seen various active substances and techniques added to the mix, including methods that use hydrogen peroxide, peracetic acid, or ozone. Just like conventional disinfection procedures, all of these methods need to be tested for efficacy. However, the established methods for testing chemical surface disinfectants do not adequately reflect how these products are being used in practice.
Generally, these kinds of processes are used as complements to "standard" surface cleaning and disinfection, and they cannot replace these methods. They may be used in areas such as isolation rooms and operating theatres, as a useful addition to the routine disinfection process.
Test Principles
In principle, gaseous agents, such as ozone, hypochlorous acid, or hydrogen peroxide, cannot be tested using previously established methods for surface disinfectants applied with a wipe, foam, or spray. In the early 1980s, a practical French standard testing method was established: the NF T 72-281 (2). This method was refined and published as DIN EN 17272 in June 2020 (3). However, the test rooms according to EN 17272 do not fully simulate such as patient rooms or operating theatres as they are not furnished for example; therefore, the test results obtained in accordance with DIN EN 17272 are not directly transferable to real-life applications.
The aim of the testing process is to obtain reproducible results to assess the general efficacy of the test and allow to compare different application conditions and procedures. For this reason, the standard recommends that additional checks be performed to verify the suitability of the test for the specific local application conditions.
Determination of Efficacy
To determine the efficacy, the test follows a practical model in which an application device is used to distribute active substances in a room. The test looks at either a combination of a device and an active substance solution or a generator that directly generates the active substance.
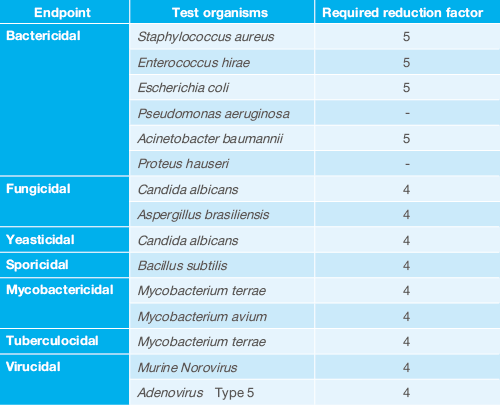
A mixture of test organisms/viruses and an organic load is applied to a stainless steel test carrier, dried, and then exposed to the disinfection process. The number of surviving test organisms or infectious virus particles is then determined and compared with the untreated control. The resulting logarithmic reduction factor (Rf) reflects the decline in living test organisms and, therefore the efficacy of the treatment. In the case of viruses, the decline in infectiousness is measured using the TCIDso (Tissue Culture Infectious Dose) process. Three process challenge devices for each process and test organism are affixed at defined heights and distances in the room so that the inoculated side is facing away from the release source.
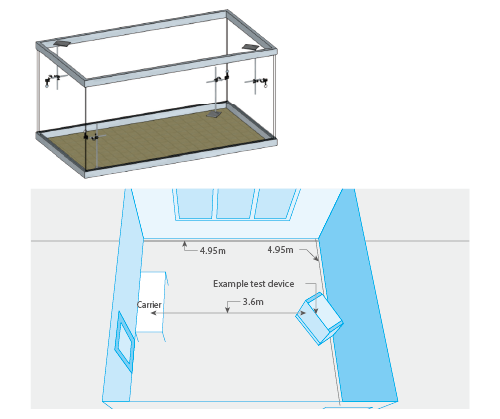
Note. Example layout of the room used for testing the air disinfection device. Carriers were placed on a table 3.6 m away from the test device, as shown. This figure is adapted from "Virucidal efficacy of an ozone-generating system for automated room disinfection" by Steinmann J et al. J Hosp Infect. 2021 Oct;116:16-20, Table 1.
In practice, a distinction is made depending on the time, which is the reaction time from the point at which the effective concentration is reached to the end of the test. In laboratory tests, the time at which the process challenge device is removed marks the end of the reaction time. The carriers can be removed as soon as the process time specified by the manufacturer has elapsed or after a decontamination phase specified by the device. Under the mandatory conditions of the standard, the reaction time must be less than 15 hours. However, this is generally a challenge for many test organisms.
Distribution Test
A new aspect of EN 17272 is the distribution test. In this test, four carriers inoculated with Staphylococcus aureus are placed in the corners of the test room: two in opposite corners, and two secured on the ceiling. In each corner, one process challenge device is positioned vertically, facing away from the source, while another is placed horizontally, facing the ceiling (if mounted on the ceiling) or the floor. In test conditions, the tested procedure must achieve a reduction of at least five log levels. The distribution test can be performed at the same time as the actual test or as a preliminary test under identical test conditions.
Conclusion and Summary
During the COVID-19 pandemic, interest in automated room disinfection procedures increased significantly. In an automated room disinfection process with ozone as the active substance, for example, tests have demonstrated efficacy against bacteriophage <1>6 and bovine coronavirus as a surrogate for SARS-CoV-24, 5.
For testing airborne chemical disinfection procedures, EN 17272 is a highly practical method, carried out in a similar way to a controlled field test. The method is a good way to standardize efficacy testing for airborne disinfection procedures and increases the safety of the disinfection as an infection prevention measure.
-
Publication Date:December 24, 2024
-
Category:Environmental Management
HosCom International 2024 Vol. 3

Other Articles in this volume
References
- Koch, R. Uber Desinfektion. Mitteilungen aus dem Kaiser!. Gesundheitsamte, 1881, Bd. I, Berlin. [Translated: Koch, R. About disinfection. Messages from the Kaiser!. Health Department, 1881, Vol. I, Berlin.]
- NF T 72-281. Methods of airborne disinfection of surfaces - Determination of bactericidal, fungicidal, yeasticidal, mycobactericidal, tuberculocidal, sporicidal and virucidal activity, including bacteriophages. November 2014. Afnor Editions; France: 2014.
- DIN EN 17272:2020-06, Chemical disinfectants and antiseptics - Methods of airborne room disinfection by automated process - Determination of bactericidal, mycobactericidal, sporicidal, fungicidal, yeasticidal, virucidal and phagocidal activities; German version EN 17272:2020. Beuth Verlag GmbH; Berlin: 2020.
- Steinmann J, Burkard T, Becker B, Paulmann D, Todt D, Bischoff B, Steinmann E, Brill FHH. Virucidal efficacy of an ozone-generating system for automated room disinfection. J Hosp Infect. 2021 Oct; 116:16-20. doi: 10.1016/j.jhin.2021.06.004.
- Franke G, Knobling B, Brill FH, Becker B, Klupp EM, Belmar Campos C, Pfefferle S, Liitgehetmann M, Knobloch JK. An automated room disinfection system using ozone is highly active against surrogates for SARS-CoV-2. J Hosp Infect. 2021 Jun; 112: 108-113, doi: 10.1016/j.jhin.2021.04.007.