A HosCom International 2024 Vol. 3 Article
SARAYA Healthcare Hygiene News
The Journey of Searching for an Emollient that Maximizes the Antibacterial Properties of CHG: The Development History of Hibiscohol A
Authors:
Dr. Koji Kihara, SARAYA Co., Ltd.
Conventional hand disinfection involves immersing both hands in a basin containing an aqueous solution of a disinfectant (Shiraishi 2016; Kato et al. 1987). However, this method has several problems, including the following:

- When the same disinfectant is used by many people, the concentration of the chemical solution in the basin decreases, weakening the disinfecting effect.
- If the chemical solution concentration decreases, there is a risk of bacterial growth in the basin.
- Long-term use of the same chemical agent can result in the emergence of resistant or tolerant bacteria.
- There is a risk of cross-infection if the same hand towels are used by multiple people after disinfecting their hands in the basin. To solve these problems, it is necessary to replace the chemicals and towels to maintain a certain level of disinfection effectiveness, which is a complicated endeavour.
Therefore, in order to obtain a simple,fast-acting, stable, and long-lasting disinfectant effect, we began developing a product that combines an alcohol-based hand disinfectant with an automatic sensor-activated dispenser to maximise the disinfectant effect of this agent.

Development history of Hibiscohol A
Here, we introduce the development history of Hibiscohol A, an alcohol-based hand rub that contains chlorhexidine gluconate (CHG) as a disinfectant.
Although a high-concentration ethanol solution of CHG has been known to have a strong disinfecting effect, it causes issues, including skin irritation and rough hands, making it difficult to use over a wide range of situations. By minimising these issues, we thought it would be possible to obtain a reliable disinfecting effect from alcohol (immediate effect), good workability (quick drying), and the sustained antibacterial effect of CHG remaining on the skin after the alcohol evaporates.
Adding an emollient is an effective way to prevent rough hands caused by high concentrations of ethanol, but if the emollient that coexists on the skin after the alcohol evaporates reduces the antibacterial activity of CHG, the durability of the antibacterial effect will be compromised and its value as a disinfectant will be halved. It is generally understood that the efficacy of disinfectants and preservatives such as CHG is affected by coexisting substances, and therefore investigating and clarifying the effect of emollients on the antibacterial activity of CHG was considered to be a fundamental and crucial issue in developing and formulating an effective hand disinfectant.
Therefore, we first selected emollients commonly used in cosmetics that had a good feel when used in an alcohol disinfectant (SFI: Skin Feel Index) (Goldenberg and De La Rosa 1971) and were soluble and stable even at low temperatures when added to 80 v/v¾ ethanol at 0.2 w/v¾ (Table 1 ). We then began to evaluate the effect of these emollients on the antibacterial activity of CHG after the alcohol had evaporated.
Specifically, we investigated various methods to evaluate the effect of emollients remaining on the skin after the evaporation of ethanol on the bactericidal activity of CHG. Various emollients were added to an 80 v/v¾ ethanol solution containing 0.2 w/v¾ CHG, and a certain amount of the solution was dropped onto filter paper. After the ethanol had evaporated, the bactericidal activity of the test bacteria was measured and compared with that of CHG alone to evaluate the effect of the emollients (Kihara and Furuta 1986).
| Table 1. Test emollients | |||
| Emollient | Trade name | Supplier | Symbol |
| Hexyl laurate | Cetiol A | Henkel hakusui | HL |
| Polyoxyethylene glyceryl monococoate | Cetiol HE | Henkel hakusui | GC |
| Propylene glycol dicaprylate | Edenol 302 | Henkel hakusui | PDC |
| Caprylic capric acid triglyceride | Myritol 318 | Henkel hakusui | CCTG |
| 2-Octyldodecanol | Eutanol G | Henkel hakusui | OD |
| Diisobutyl adipate | Vinyeizer 40 | Kao | DIBA |
| Glyceryl tri (2-ethylhexanoate) | Exceparl TGO | Kao | GTEH |
| Isopropyl myristate | IPM | Henkel hakusui | IPM |
| Isopropyl palmitate | Nikkol IPP | Nikko chemical | IPP |
| Isopropyl isotearate | Nikkol IPIS | Nikko chemical | IPIS |
| Cetyl lactate | Nikko chemical | CL | |
| Diisopropyl adipate | Nikkol DID | Nikko chemical | DIPA |
| Polyglycerol | Polyglycerin #500 | Sakamoto yakuhin kogyo | PG |
The test organisms used in the study were S. aureus and P. aeruginosa, and 13 types of emollients were tested. Interestingly, we found that diisobutyl adipate (DIBA) and polyoxyethylene glyceryl monococoate (GC) enhanced the bactericidal activity of CHG (Fig. 1 & 2).
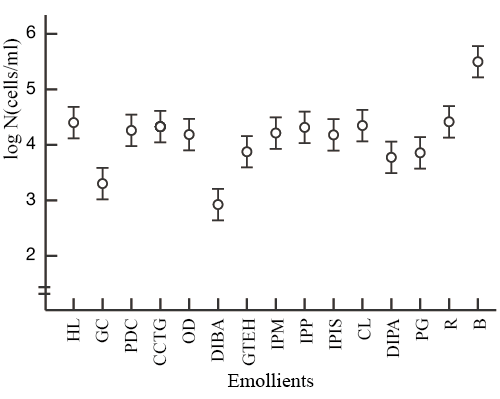
|
Test solution: 80v/v% ethanol + 0.2w/v% chlorhexidine
digluconate + 0.2w/v% emollient
R: 80v/v% ethanol + 0.2w/v% chlorhexidine digluconate
B: 80v/v% ethanol
Markers have a 95% confidence interval
|
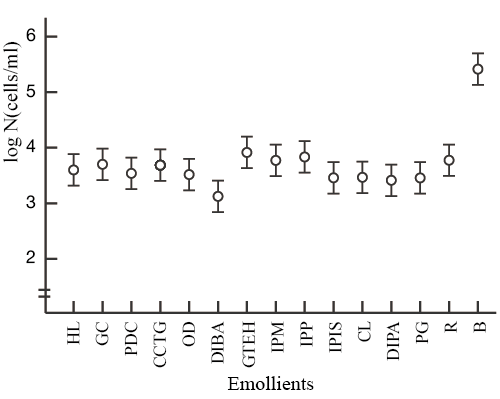
|
Test solution: 80v/v% ethanol + 0.2w/v% chlorhexidine
digluconate + 0.2w/v% emollient
R: 80v/v% ethanol + 0.2w/v% chlorhexidine digluconate
B: 80v/v% ethanol
Markers have a 95% confidence interval
|
Furthermore, when the effect of varying the concentration of DIBA in 80 v/v¾ ethanol containing 0.2 w/v¾ CHG on the viable cell count of S. aureus was examined, the addition of 0.1 w/v¾ DIBA significantly enhanced the bactericidal activity of CHG (Fig. 3), and an ethanol solution of 0.2 w/v¾ CHG containing 0.1 w/v¾ DIBA showed the same bactericidal effect as 0.4 w/v¾ CHG alone.
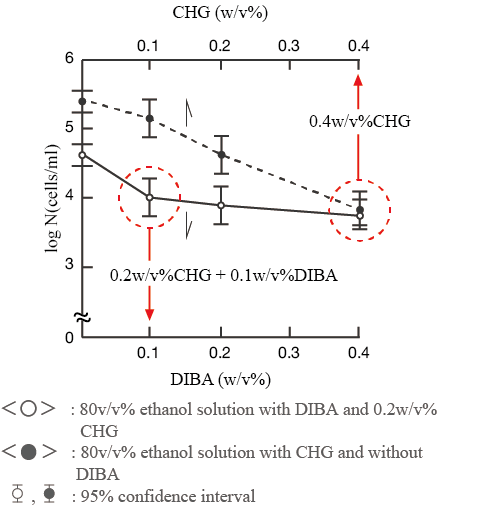
Based on the results, Hibiscohol A has been commercialised.
-
Publication Date:December 24, 2024
-
Category:Hand Hygiene
HosCom International 2024 Vol. 3

Other Articles in this volume
References
- Shiraishi, T. (2016) 'Correct use of disinfectants-based on the history of disinfectants', Japanese Journal of Environmental Infections, vol. 31, no. 4, pp. 224-229.
- Kato, M., et al (1987) 'The reasonable time of exchange of the disinfectant solution in a basin on 0.02% Hibitane (chlorhexidine) solution', Journal of Japanese Society of Nursing Research, vol. 9, no. 4, pp. 64-71.
- Goldenberg, R., and De La Rosa. (1971) 'Correlation of skin feel of emollients to their chemical structure', Journal of the Society of Cosmetic Chemists, vol. 22, pp. 635-654.
- Kihara, K., and Furuta, T. (1986) 'Effect of various emollients on the bactericidal activity of chlorhexidine digluconate', Journal of Antibacterial and Antifungal Agents, vol. 14, no. 12, pp. 597-604.